Imaging
Our imaging facility is designed to respond to the current and future needs of developmental biology and cell biology in whole living organisms. Our imaging systems are either custom made or custom adapted from commercial setups (Leica, LaVisionBiotech).
Click here if you want to know more about multiphoton microscopy.
Our setups for in vivo multiscale microscopy imaging
Our imaging setups are mainly located in Gif-sur-Yvette building 32. In addition, we installed part of our equipement (including a Leica SP8 setup coupled with an Insight femtosecond laser) in the Observatoire Océanologique of Banyuls-sur-Mer (OOB, Oceanological Observatory of Banyuls-sur-Mer).
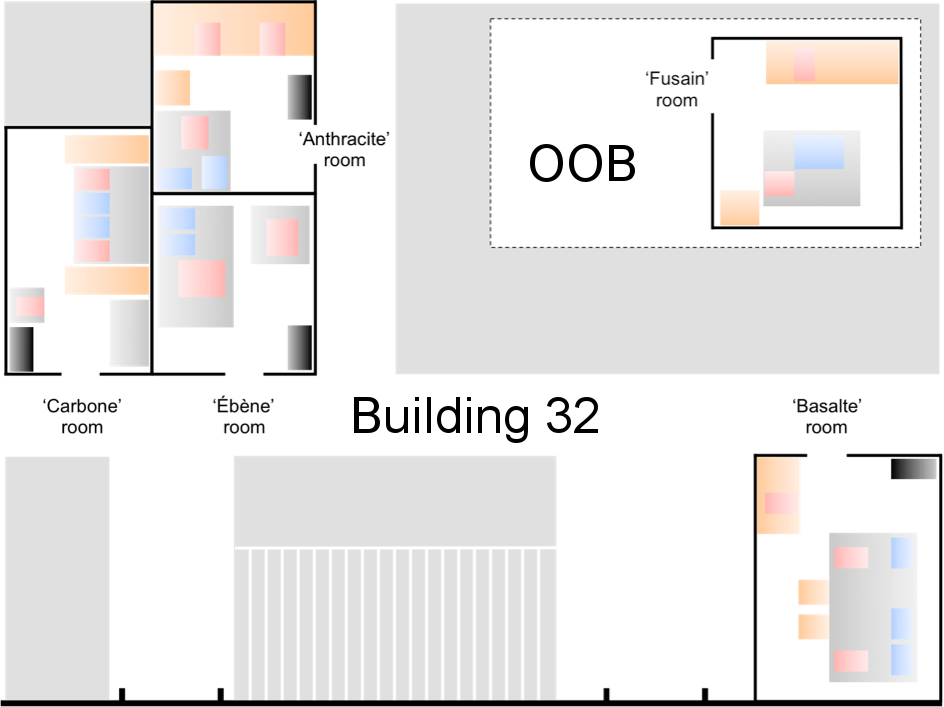
Map of the rooms in building 32 (Gif-sur-Yvette) and OOB (Banyuls). Red: microscopes; Blue: lasers; Orange: desks and lab benches; Black: UPS
Room Carbone (Building #32, ground floor)
Two setups (Leica SP5) are coupled to two femtosecond lasers (Mai Tai Spectra Physics and t-pulse 20 Amplitude Systems) through an original optical bench (Designed by Pierre Suret PHLAM Lille). The two setups thus offer similar imaging conditions and are optimized for both excitation (by the two IR lasers) and detection (Hybrids ultrasensitive detectors) of commonly used fluorophores. A temperature and gas controlling system is available on both systems (Okolab).
 |
Leica SP5 #1 |
|---|---|
|
|
 |
Leica SP5 #2 |
|
|
 |
Light-sheet microscope |
|
|
 |
Leica stereomicroscope for specimen mounting |
|
Room Anthracite (Building #32, ground floor)
All-in-one Zeiss confocal (LSM780) and two-photon microscope with spectral detection, fully motorized stage and temperature and gas controller (Okolab). This setup is more particularly dedicated to multicolor imaging (e.g. for multicolor ISH or Brainbow type staining) on large specimens (3D mosaic imaging).
Nomarski video-microscope (Olympus BX51) equipped with water dipping lens objectives. It provides high quality images and movies of your favorite transparent specimens. It is also equipped with temperature control (Okolab).
Room Basalte (Building #32, ground floor)
Two Lavision Biotech two-photon microscopes coupled with three IR femtosecond lasers, enabling optimized excitation of multiple fluorophores. Both setups are equipped with multichannel detection in both reflection and transmission and give the possibility to record SHG and THG signals in combination with fluorescence signals. The systems are open (both hardware and software) and allow us to develop and implement the artificial assistant concept. The aim is to analyze in real time relevant features in order to:
- provide a feedback control on acquisition parameters according to variation of signals
- recognize and follow patterns of interest
- alternate different imaging modes such as large field of view and close up on ROI
Room Ebène (Building #32, ground floor)
Development of custom made light sheet microscopes (collaboration with Pierre Suret PHLAM Lille, Pablo Loza ICFO Barcelone and partnership with PhaseView). We currently build a new evolving light sheet microscope (SPIM and then DSLM, one-photon and then two-photon excitation, single and then two-sided excitation...). The software is developed in Python to be compatible with the artificial assistant. Development of a setup dedicated to bioluminescence imaging based on an Olympus BX51 microscope.
Room Fusain (OOB, Banyuls-sur-Mer)
Partnership with OOB for the exploration of marine embryonic biodiversity. Leica SP8 two-photon microscope coupled with a tunable Spectraphysics Insight laser. The latter covers a broad range of wavelengths (720-1300nm) and gives us the possibility to acquire both fluorescence and harmonic signals (SHG and THG). In the context of our project SeaLim (live imaging of marine model organisms), this setup was first operating in the marine biology station of Roscoff during the summer 2013 and in the Observatoire Océanologique of Villefranche-sur-Mer from April 2014 to March 2015.
 |
Leica SP8 |
|---|---|
|
|
 |
Leica stereomicroscope for specimen mounting |
|
Choice of the microscope objective
The main important characteristics that should be taken into account
Objective choice depends on the answers to the following questions :
- What is the size of the specimen or region of interest?
- Determine the size of the field of view you need and hence the magnification of the objective.
- What is the smallest object to be identified?
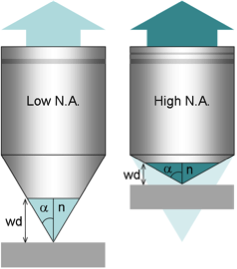
- Compare the size of the objects with the theoretical resolution achieved with a certain NA and wavelength.
- What is the brightness of the labeling and what is the sensitivity of the sample to light?
- A greater NA, even if not used for reaching a very small resolution is also important for collecting photons since the angle of photon collection is greater.
- At what wavelength will the experiment be performed?
- Find help in the wavelength excitation section and take care that the objective you selected is efficient for transmitting such the chosen wavelength
Main characteristics of our microscope objectives
All our objectives can be used on any of our microscopes. In case, we have a collection of adaptation rings.
We can extend our collection of objectives to respond to your needs
| Brand | Mag. | NA | WD | Immersion | Transmission curve | Picture |
|---|---|---|---|---|---|---|
| Zeiss | 5x | 0.16 | 12.1 | Air | 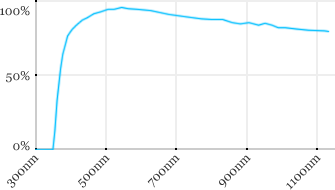 |
 |
| Leica | 10x | 0.3 | 3.6 | Water |  |
|
| Zeiss | 10x | 0.3 | 2.6 | Water | 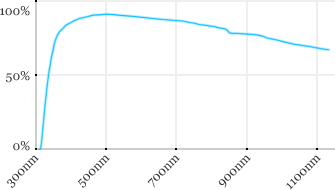 |
 |
| Zeiss | 10x | 0.45 | 1.8 | Water | 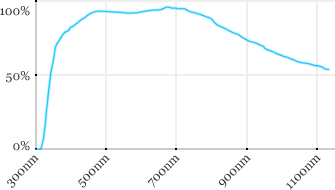 |
 |
| Nikon | 16x | 0.8 | 3 | Water |  |
|
| Leica | 20x | 0.5 | 3.5 | Water | 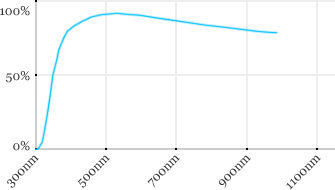 |
 |
| Olympus | 20x | 0.5 | 3.5 | Water | 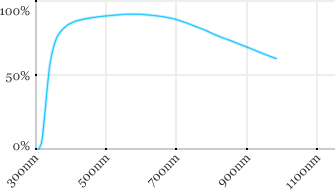 |
 |
| Zeiss | 20x | 0.5 | 2.6 | Water | 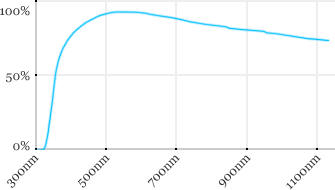 |
 |
| Olympus | 20x | 0.95 | 2 | Water | 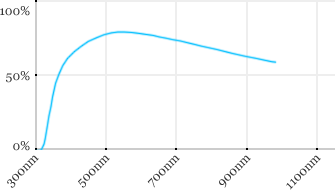 |
 |
| Zeiss | 20x | 1 | 1.8 | Water | 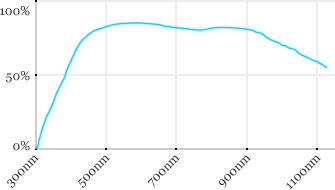 |
 |
| Leica | 20x | 1 | 1.95 | Water |  |
|
| Nikon | 25x | 1.1 | 2 | Water |  |
|
| Leica | 40x | 0.8 | 3.3 | Water | 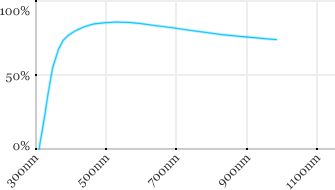 |
 |
| Olympus | 40x | 0.8 | 3.3 | Water | 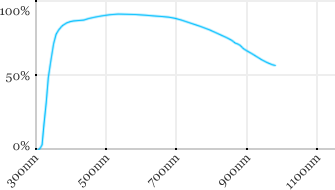 |
 |
| Zeiss | 40x | 0.8 | 2.1 | Water | 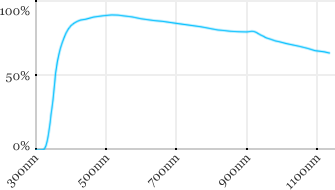 |
 |
| Zeiss | 40x | 1 | 2.5 | Water | 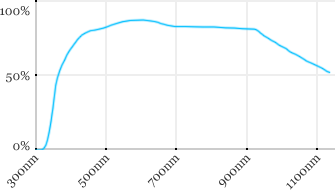 |
 |
| Olympus | 60x | 1.1 | 1.5 | Water | 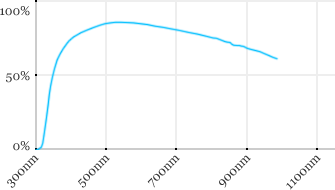 |
 |
| Leica | 63x | 0.9 | 2.2 | Water | 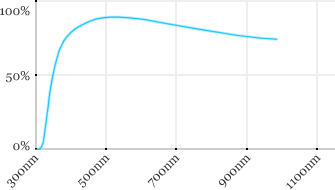 |
 |
| Zeiss | 63x | 0.9 | 2.4 | Water | 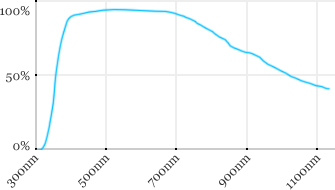 |
 |
| Zeiss | 63x | 1 | 2.1 | Water | 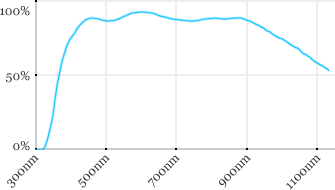 |
 |
Measured transmission in the infrared range
Measurements done on the SP8/DM6000 Leica stand, with an Insight laser (Spectra-Physics) with a series of long working distance high-NA objectives.